For the reaction N2O4(g) ⇌ 2NO2(g), Kp = 0.492atm at 300K. Kc for the reaction at same temperature is × 10−2.
(Given : R = 0.082L atmmol−1K−1)
[29-Jan-2024 Shift 1]
The following concentrations were observed at 500K for the formation of NH3 from N2 and H2. At equilibrium :
[N2] = 2 × 10−2M, [H2] = 3 × 10−2M and [NH3] = 1.5 × 10−2M. Equilibrium constant for the reaction is___
[29-Jan-2024 Shift 2]
For the given reaction, choose the correct expression of KC from the following :-
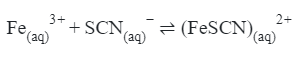
[31-Jan-2024 Shift 1]
Given below are two statements :
Statement (I) : Aqueous solution of ammonium carbonate is basic.
Statement (II) : Acidic/basic nature of salt solution of a salt of weak acid and weak base depends on Ka and Kb value of acid and the base forming it.
In the light of the above statements, choose the most appropriate answer from the options given below :
[27-Jan-2024 Shift 1]
Which of the following is strongest Bronsted base?
[27-Jan-2024 Shift 1]
The pH at which Mg(OH)2[K sp = 1 × 10−11] begins to precipitate from a solution containing 0.10M Mg2+ ions is____
[30-Jan-2024 Shift 1]
The pH of an aqueous solution containing 1M benzoic acid (pKa = 4.20) and 1M sodium benzoate is 4.5. The volume of benzoic acid solution in 300 mL of this buffer solution is_____ mL.
[30-Jan-2024 Shift 2]
Ka for CH3COOH is 1.8 × 10−5 and Kb for NH4OH is 1.8 × 10−5. The pH of ammonium acetate solution will be
[1-Feb-2024 Shift 1]
Solubility of calcium phosphate (molecular mass, M) in water is Wg per 100mL at 25∘C. Its solubility product at 25∘C will be approximately.
[1-Feb-2024 Shift 2]









 ×
×