 192:59 MIN
192:59 MINChoose the polar molecule from the following :
[27-Jan-2024 Shift 1]
Sum of bond order of CO and NO+ is____
[27-Jan-2024 Shift 1]
Number of compounds with one lone pair of electrons on central atom amongst following is O3, H2O, SF4, ClF3, NH3, BrF5, XeF4 __
[29-Jan-2024 Shift 1]
The number of species from the following which are paramagnetic and with bond order equal to one is____
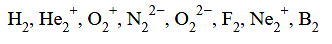
[29-Jan-2024 Shift 1]
The total number of molecules with zero dipole moment among CH4, BF3, H2O, HF, NH3, CO2 and SO2 is______
[29-Jan-2024 Shift 2]
The total number of 'Sigma' and Pi bonds in 2formylhex-4-enoic acid is
[29-Jan-2024 Shift 2]
The total number of anti bonding molecular orbitals, formed from 2 s and 2p atomic orbitals in a diatomic molecule is___
[29-Jan-2024 Shift 2]
Match List-I with List-II
| List-I Molecule |
List-II Shape |
|---|---|
| (A) BrF5 | (I) T-shape |
| (B) H2O | (II) See saw |
| (C) ClF3 | (III) Bent |
| (D) SF4 | (IV) Square pyramidal |
[30-Jan-2024 Shift 1]
The total number of molecular orbitals formed from 2 s and 2p atomic orbitals of a diatomic molecule
[30-Jan-2024 Shift 1]
Given below are two statements:
Statement-I: Since fluorine is more electronegative than nitrogen, the net dipole moment of NF3 is greater than NH3.
Statement-II: In NH3, the orbital dipole due to lone pair and the dipole moment of NH bonds are in opposite direction, but in NF3 the orbital dipole due to lone pair and dipole moments of N-F bonds are in same direction.
In the light of the above statements. Choose the most appropriate from the options given below.
[30-Jan-2024 Shift 2]
The linear combination of atomic orbitals to form molecular orbitals takes place only when the combining atomic orbitals
A. have the same energy
B. have the minimum overlap
C. have same symmetry about the molecular axis
D. have different symmetry about the molecular axis
Choose the most appropriate from the options given below:
[31-Jan-2024 Shift 1]
The number of species from the following in which the central atom uses sp3 hybrid orbitals in its bonding is______
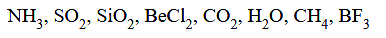
[31-Jan-2024 Shift 1]
Which of the following is least ionic?
[31-Jan-2024 Shift 2]
A diatomic molecule has a dipole moment of 1.2D. If the bond distance is 1Å, then fractional charge on each atom is ____ × 10−1 esu.
(Given 1D = 10−18 esu cm)
[31-Jan-2024 Shift 2]
Arrange the bonds in order of increasing ionic character in the molecules. LiF, K2O, N2, SO2 and CIF3
[1-Feb-2024 Shift 1]
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : PH3 has lower boiling point than NH3.
Reason (R) : In liquid state NH3 molecules are associated through vander waal's forces, but PH3 molecules are associated through hydrogen bonding.
In the light of the above statements, choose the most appropriate answer from the options given below:
[1-Feb-2024 Shift 1]
The number of molecules/ion/s having trigonal bipyramidal shape is ........ .
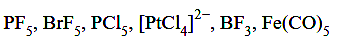
[1-Feb-2024 Shift 1]
Given below are two statements :
Statement (I) : A π bonding MO has lower electron density above and below the inter-nuclear asix.
Statement (II) : The π* antibonding MO has a node between the nuclei.
In the light of the above statements, choose the most appropriate answer from the options given below:
[1-Feb-2024 Shift 2]
Select the compound from the following that will show intramolecular hydrogen bonding.
[1-Feb-2024 Shift 2]












 ×
×